Cosa sono i passaggi di stato
In questo articolo trovate una spiegazione relativa a cosa sono i passaggi di stato (o cambiamenti di stato).
Come abbiamo potuto vedere nell’articolo precedente ogni sostanza, in condizioni normali si presenta in un determinato stato di aggregazione: solido, liquido o aeriforme.
Tuttavia la sostanza sottoposta a riscaldamento o raffreddamento può passare da uno stato all’altro, può quindi subire passaggi di stato, vediamoli in dettaglio:
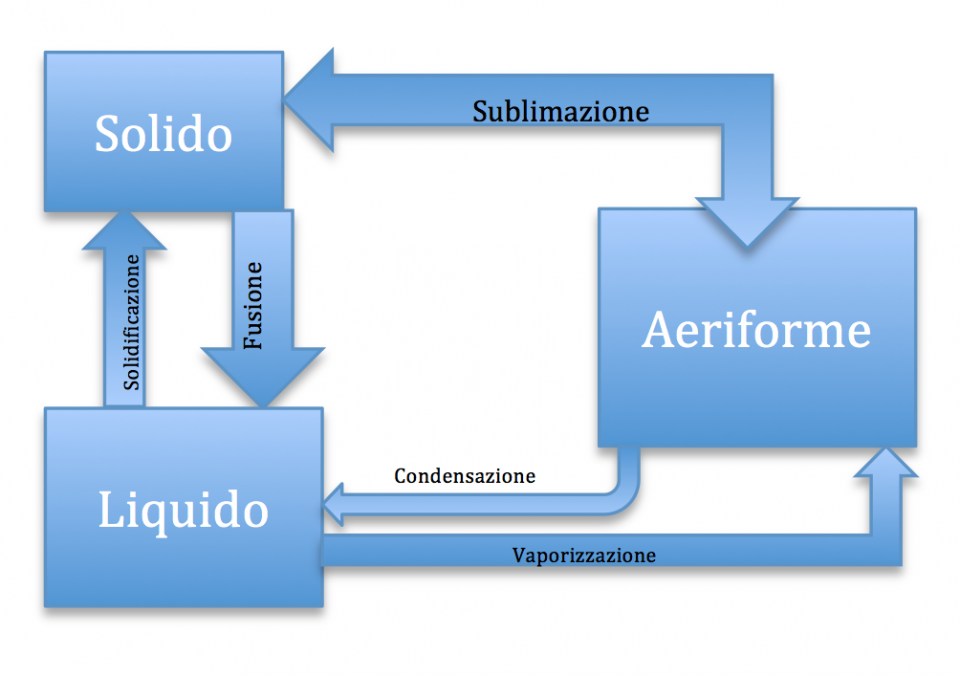
Il passaggio dallo stato liquido a quello solido si chiama solidificazione.
Il passaggio dallo stato solido a quello liquido si chiama fusione.
Il passaggio dallo stato aeriforme allo stato solido è detto sublimazione.
Mentre il passaggio dallo stato liquido a quello aeriforme prende il nome di vaporizzazione.
Il passaggio dallo stato aeriforme allo stato liquido è chiamato condensazione.
Per riassumere riporto qui di seguito uno schema riassuntivo:
Al variare della temperatura le sostanze possono dunque passare da uno stato di aggregazione all’altro. Questo perché l’aumento della temperatura modifica l’agitazione termica e la forza di coesione delle molecole.
Con più temperatura le molecole acquistano maggior capacità di movimento poiché la loro forza di coesione diventa sempre più debole.
Ecco i cambiamenti di stato nella vita di tutti i giorni:
Se lasciate una pentola d’acqua sulla piastra dopo un po’ di tempo risulterà vuota, questo perché l’acqua da liquida è diventata aeriforme, ovvero vapore acqueo.
Il vapore acqueo del tuo respiro a contatto con il vetro freddo di un finestrino o di una finestra in generale, lo appanna; è quindi possibile affermare che il vapore acqueo dallo stato aeriforme è passato allo stato liquido, visibile grazie alle goccioline presenti sul finestrino.
